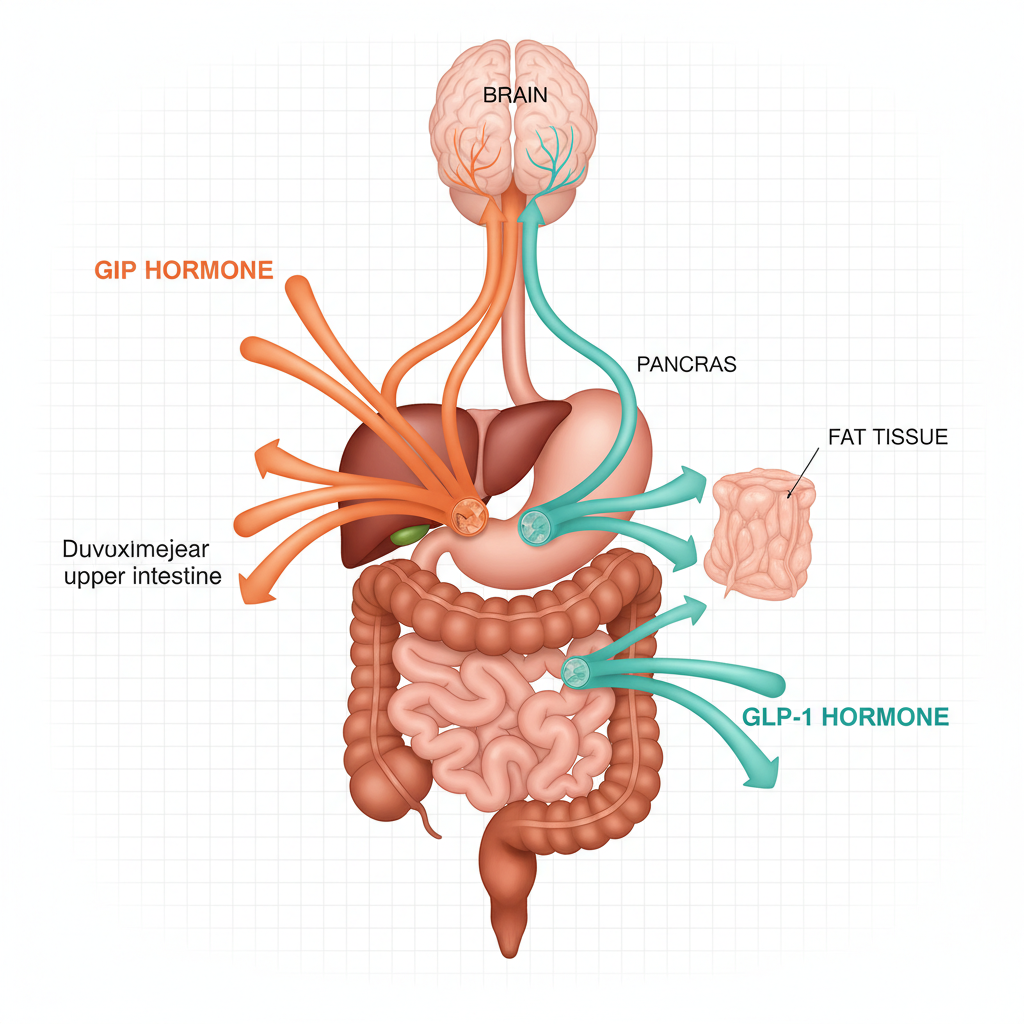
Incretin hormones GLP-1 and GIP play a central role in regulating appetite, glucose metabolism, and body weight. Understanding their function helps to better grasp why tirzepatide (Mounjaro), which simultaneously targets both hormones, achieves superior results compared to mono-agonist treatments.
Incretins: essential digestive hormones
The concept of incretins emerged from a major clinical observation: oral glucose administration causes significantly higher insulin secretion than that obtained by intravenous injection of the same amount of glucose. This incretin effect, discovered in the 1960s, accounts for 50 to 70% of the total postprandial insulin response.
Two hormones are responsible for this effect: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Secreted by specialized intestinal cells in response to food intake, they orchestrate a cascade of metabolic signals that extend far beyond simple blood glucose regulation.
GLP-1: the satiety hormone
GLP-1 is synthesized by L-cells of the distal ileum and colon from proglucagon, a peptide precursor. Its secretion begins 10 to 15 minutes after the start of a meal, with a peak reached between 30 and 60 minutes.
Mechanisms of GLP-1 action:
On the pancreas: GLP-1 binds to its receptor (GLP-1R) on pancreatic beta cells, stimulating insulin secretion in a glucose-dependent manner. This means that the insulin-secretory effect only manifests when blood glucose is elevated, minimizing the risk of hypoglycemia. It also inhibits glucagon secretion by alpha cells, reducing hepatic glucose production.
On the brain: GLP-1 receptors are present in several brain regions involved in appetite control: the nucleus of the solitary tract (NTS) in the brainstem, the arcuate nucleus of the hypothalamus, and the area postrema. Activation of these receptors reduces hunger signals and increases feelings of satiety. GLP-1 also modulates the dopaminergic reward system, decreasing the appeal of high-fat and high-sugar foods (Turton et al., Nature, 1996).
On the stomach: GLP-1 significantly slows gastric emptying, which prolongs stomach distension and mechanical satiety signals. This effect partly explains the nausea observed at the beginning of GLP-1 agonist treatment.
On the cardiovascular system: GLP-1 receptors are expressed on cardiomyocytes and endothelial cells. GLP-1 improves endothelial function, reduces vascular inflammation, and has demonstrated cardioprotective effects in major cardiovascular studies (LEADER, SUSTAIN-6, SELECT).
Natural half-life of GLP-1: only 2 to 3 minutes. The enzyme DPP-4 (dipeptidyl peptidase-4) rapidly degrades GLP-1 into inactive metabolites. This ultra-rapid degradation necessitated the development of resistant forms (GLP-1 analogues) or DPP-4 inhibitors.
GIP: the overlooked metabolic hormone
GIP is secreted by K-cells of the duodenum and proximal jejunum. Its secretion is primarily triggered by the absorption of glucose and lipids, with a plasma peak reached 15 to 30 minutes after a meal.
Long considered a minor hormone in metabolic physiology, GIP has been undergoing a complete reevaluation since the work of Muller et al. (Nature Reviews Drug Discovery, 2022).
Mechanisms of GIP action:
On the pancreas: GIP is the most potent stimulator of insulin secretion among incretins, responsible for approximately 60% of the total incretin effect. Like GLP-1, its action is glucose-dependent. It also stimulates the proliferation and survival of pancreatic beta cells.
On adipose tissue: This is where GIP fundamentally differs from GLP-1. GIP receptors (GIP-R) are highly expressed in white adipose tissue. Activation of GIP-R in adipose tissue produces complex and context-dependent effects:
- Under normal physiological conditions: GIP promotes lipid storage and adipogenesis
- Under sustained pharmacological activation (as with tirzepatide): it promotes lipolysis, fat mobilization, and browning of white adipose tissue
- It improves insulin sensitivity in adipose tissue
- It reduces chronic adipocyte inflammation associated with obesity
On the brain: GIP receptors are present in the hypothalamus, cerebral cortex, and hippocampus. GIP modulates appetite through pathways distinct from those of GLP-1, acting notably on food motivation and reward circuits. Studies in mice show that activation of GIP-R neurons in the hypothalamus reduces food intake independently of GLP-1 (Kaneko et al., Cell Metabolism, 2023).
On bones: GIP stimulates bone formation by activating osteoblasts and partially inhibiting bone resorption. This is why tirzepatide may have an advantage over pure GLP-1 agonists for preserving bone density during weight loss.
Natural half-life of GIP: approximately 5 to 7 minutes, also degraded by DPP-4.
The difference between healthy and diabetic patients
A paradox long hindered the development of GIP-based therapies: in patients with type 2 diabetes, the insulin response to GIP is severely impaired. The incretin effect is reduced by 80% compared to healthy subjects.
| Parameter | Healthy Subject | Type 2 Diabetes |
|---|---|---|
| Total incretin effect | 50-70 % | 20-30 % |
| GIP Response | Normal | Severely reduced |
| GLP-1 Response | Normal | Partially preserved |
| GLP-1 Secretion | Normal | Slightly reduced |
| GIP Secretion | Normal | Normal or increased |
Source: Nauck MA, Meier JJ. Diabetologia. 2011;54(1):10-18.
This paradox was resolved by the discovery that supraphysiological pharmacological activation of the GIP receptor (such as that produced by tirzepatide) can restore GIP sensitivity in diabetic patients, even surpassing the normal physiological response.
GLP-1 + GIP Synergy: why 1 + 1 = 3
The combination of the two hormones produces synergistic effects that go beyond the simple addition of their individual actions:
1. On weight loss: GLP-1 reduces appetite via the brainstem and slows gastric emptying. GIP activates complementary pathways in the hypothalamus and acts directly on adipose tissue. Result: weight loss with tirzepatide (22.5%) is 5 to 6 percentage points higher than with semaglutide (16.9%).
2. On glycemic control: Dual insulin-secretory stimulation potentiates the pancreatic response. HbA1c reduction is 2.07% with tirzepatide 15 mg, compared to 1.86% with semaglutide 2 mg.
3. On body composition: GIP promotes fat mass loss while preserving lean mass. The SURMOUNT-1 study showed that 85% of weight loss with tirzepatide came from fat mass, a higher ratio than observed with semaglutide.
4. On tolerability: Tirzepatide’s signaling bias on the GLP-1 receptor (favoring the cAMP pathway over beta-arrestin) reduces gastrointestinal effects. Nausea is generally less severe and shorter-lived than with semaglutide.
Current and future therapeutic applications
Understanding incretin physiology has led to the development of four classes of medications:
- DPP-4 inhibitors (sitagliptin, vildagliptin): prevent the degradation of natural incretins. Modest effects on weight and blood glucose.
- GLP-1 receptor agonists (semaglutide, liraglutide, dulaglutide): mimic GLP-1 action with prolonged duration.
- Dual GIP/GLP-1 agonist (tirzepatide): the first molecule to activate both receptors simultaneously.
- Triple GIP/GLP-1/glucagon agonist (retatrutide, in phase 3): adds the glucagon receptor to further increase energy expenditure.
Key takeaways
GLP-1 and GIP are two complementary hormones that, together, control more than half of the insulin response after a meal. While GLP-1 primarily acts on appetite, gastric emptying, and the cardiovascular system, GIP plays a unique role in adipose tissue metabolism and bone formation. Tirzepatide leverages this complementarity to produce clinical results superior to treatments targeting a single hormone.
FAQ
What is the difference between GLP-1 and GIP?
GLP-1 primarily acts on appetite (via the brain), gastric emptying, and the pancreas. GIP targets adipose tissue, bone metabolism, and pancreatic insulin response more. Both are incretin hormones secreted by the intestine after a meal.
Why was GIP long overlooked in treatments?
In diabetic patients, the natural response to GIP is severely impaired. Researchers therefore thought that targeting GIP would be ineffective. Tirzepatide proved otherwise: pharmacological activation at supraphysiological doses restores and amplifies GIP’s effects.
Does natural GLP-1 last long in the body?
No. Natural GLP-1 has a half-life of only 2 to 3 minutes before being degraded by the DPP-4 enzyme. This is why GLP-1 agonist medications are chemically modified to resist this degradation and last for several days.
Does tirzepatide stimulate both hormones or replace them?
Tirzepatide does not stimulate the natural secretion of GLP-1 and GIP. It replaces their action by directly activating their cellular receptors, like a key that would open two different locks simultaneously.
Are DPP-4 inhibitors as effective as agonists?
No. DPP-4 inhibitors (like sitagliptin) prevent the degradation of natural incretins but only increase their levels by 2 to 3 times. Agonists like tirzepatide reach concentrations 10 to 100 times higher than physiological levels, producing much more powerful effects on weight and blood glucose.
Sources
- Nauck MA, Meier JJ. The incretin effect in healthy individuals and those with type 2 diabetes. Diabetologia. 2011;54(1):10-18.
- Muller TD et al. GIP and GLP-1 as therapeutic targets in diabetes and obesity. Nat Rev Drug Discov. 2022;21:513-532.
- Turton MD et al. A rôle for glucagon-like peptide-1 in the central régulation of feeding. Nature. 1996;379:69-72.
- Kaneko K et al. GIP receptor signaling in neurons mediates body weight réduction. Cell Metabolism. 2023.
- Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab. 2013;17(6):819-837.
- Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev. 2007;87(4):1409-1439.